Don’t disturb the waves, eliminate the particles
Noli tangere circulos meos, ” or “don’t touch my particles,” Archimedes of Syracuse supposedly said just before he was killed by a Roman soldier’s spear. We can only speculate about what he truly meant. Still, he captured quite well how the quantum world seems to perceive our attempts to understand it, along with their consequences.
If we throw a stone into the water, circular ripples begin to spread across the otherwise calm surface. These circular waves will rock a duck floating on the water. However, the duck won´t move from its place. The waves don´t carry any mass, only energy. The vibrating duck partially absorbs energy from the waves. This disturbs them and results in a slight damping of the ripples.
Disturbed Ripples
We encounter the same image in the physics of quantum systems, except that “quantum waves” are a somewhat more abstract object. Instead of carrying energy, they carry probabilities. Not just one probability, but the probabilities of everything we can observe – probabilities of energy, probabilities of velocity, probabilities of position, and so on. Quantum waves themselves are not directly visible, and we cannot touch them with our senses. We know of their existence through instruments, which, like the duck on the water, wait until they disturb the entire quantum system.

Young’s double-slit experiment: particle vs. wave
However, there is one more important difference. Imagine ten ducks floating on the surface, all equally distant from the point where a “stone” splashed and created a quantum system. In the case of water waves, we would expect all ten ducks to rock. On a quantum surface, however, only one duck rocks. Which one? That is the result of quantum randomness. If we repeated the experiment 1000 times, each duck would rock roughly 100 times.
When a quantum wave is disturbed, the original wave either disappears or transforms into a completely different wave. For example, a photon, described as a quantum wave, when passing through a polarizer, either vanishes completely or continues as a quantum wave with a specific polarization. The resulting polarization, however, has nothing to do with the photon’s original polarization. Disturbing the quantum wave is therefore a far more dramatic act than simply rocking an object on the water’s surface. The measurement process literally ‘materializes’ the properties, introducing an element of true randomness.
Light versus Slits
In the 18th century, Isaac Newton and Christiaan Huygens argued about the nature of light. Newton considered light to be a stream of particles, while Huygens thought of it as a wave. In 1801, Thomas Young performed one of the most famous experiments in physics – the double-slit experiment, to experimentally determine who was correct. He placed a barrier with two narrow, closely spaced slits in the path of monochromatic light. The light passed through the slits, and on the photographic plate positioned behind the barrier, he observed where the light landed.
If Isaac Newton was right, the resulting illumination on the plate would be the sum of the illuminations that would occur if only one of the slits were open. In other words, the counts of particles passing through the first or second slit would add together, producing two overlapping bands of light shifted relative to the slit separation.
According to Huygens’ theory, each slit acts as a source of waves. Behind the barrier, two semicircular light waves are created, and where they meet, they combine like ordinary waves. Some areas are reinforced while others cancel out. Instead of two overlapping light bands, the plate shows alternating bright and dark stripes, creating what we now call an interference pattern.
Waves, Ripples, Particles
Thomas Young’s experiment clearly demonstrated that light has a wave nature. So why do we still talk about photons today? If we look closely at an interference pattern, at the molecular level we see that it is made up of countless dots – traces left on the photographic plate by individual absorbed photons.According to quantum physics, a photon propagates through space as a quantum wave that carries the probability of its occurrence, until it appears (with a certain probability) at a particular spot on the photographic plate. Light-sensitive molecules then use its energy in a chemical process, which causes the photographic plate to darken at that location.The photon thus has a wave nature. Louis de Broglie was the first to propose that the photon is not unique in this respect, and that every quantum particle, every quantum system, has a wave nature. In his 1924 doctoral dissertation, he described the wave nature of electrons. He assigned a quantum wave to the electron, with a wavelength λ = h/p, where h=6.62607015 x 10-34 J.s-1 is Planck’s constant, and p is the electron’s momentum. At room temperature, free electrons (mass 9.1 x 10-31 kg) in conductive materials move at speeds of roughly 107 m/s. Their corresponding quantum waves have wavelengths on the order of nanometers, comparable to photons of X-ray (RTG) radiation. A charge with a mass of 10 g moving at 1 000 m/s has a wavelength on the order of 10-26 nm, which is smaller than the Planck scale and considered unmeasurable. The most energetic gamma-ray photons observed so far (originating from cosmic radiation) have a wavelength of about 10-21 nm.
Images of the World X
The double-slit experiment is conceptually the simplest type of interference experiment –waves passing through a medium with obstacles. The structural arrangement and properties of the obstacles can be identified from the resulting interference pattern. A crystal lattice is another example of such a medium: regularly arranged atoms of elements forming, for instance, table salt or diamond. In February 1912, physicist Max von Laue came up with the idea of using X-ray radiation, whose wavelength corresponds to the expected distances between atoms, to test the hypothesis of a crystal structure.
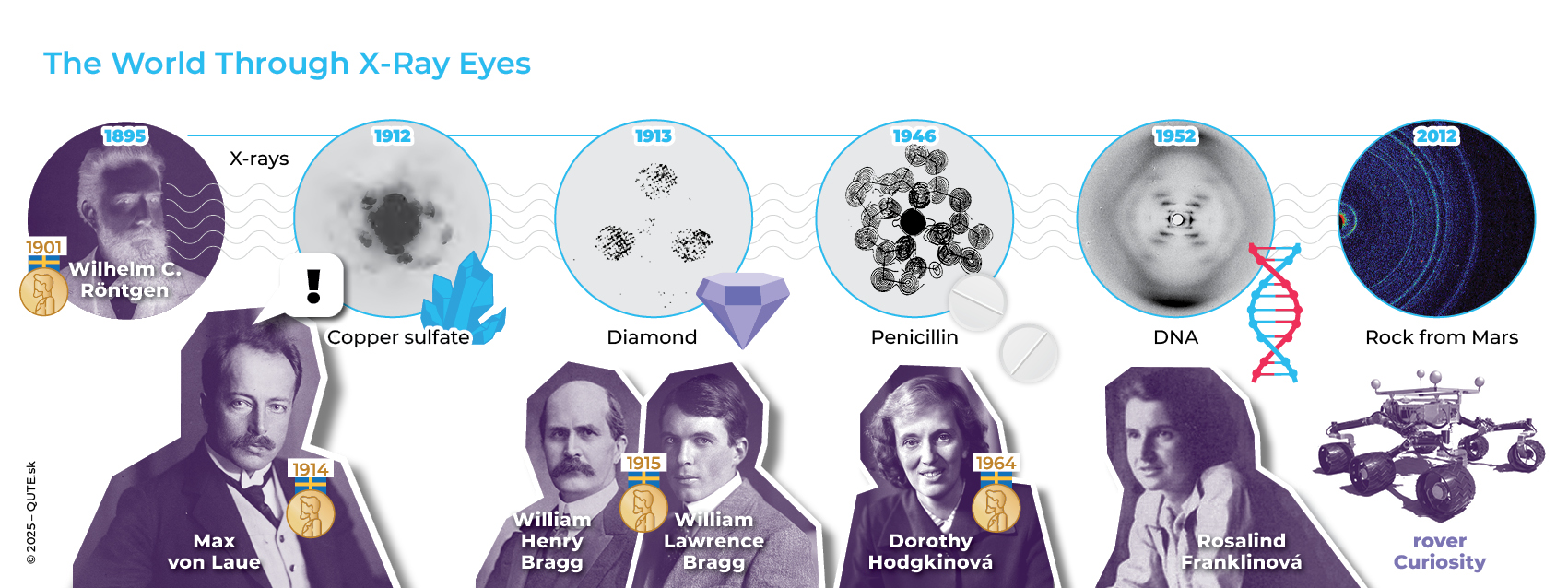
Thanks to the X-rays discovered by W. C. Röntgen and Max von Laue’s idea, we can now study the structure of matter directly.
The experiment was carried out with the help of Paul Knipping and Walter Friedrich in the same year. They were the first to observe a diffraction interference pattern for a crystal of copper (II) sulfate (CuSO4). This experiment confirmed the wave nature of X-rays, which had been debated for 17 years since their discovery, and at the same time introduced a technology that allows us to look inside matter.Later, William Henry Bragg and William Lawrence Bragg, father and son, significantly improved the methods and theory. Their work on revealing the structure of diamond helped explain its hardness. One crystal after another reveals its face under X-ray light. Beyond studying the structure of materials and molecules, X-ray diffraction has revealed many properties of chemical bonds. Chemist Dorothy Hodgkin applied X-ray crystallography methods to biological molecules, revealing the structures of cholesterol, penicillin, and insulin. Using X-ray diffraction, Rosalind Franklin confirmed the double-helix structure of DNA in 1952.
Interference of Matter
When studying the structure of matter, X-ray radiation can be replaced by electrons, which have a similar wavelength. The wave nature of electrons was independently demonstrated in two diffraction experiments in 1927. Clinton Davisson and Lester Germer observed the diffraction of electrons on a nickel crystal, while George P. Thomson (son of the discoverer of the electron as a particle) and his doctoral student Alexander Reid observed electron diffraction on thin metal foils of aluminum, gold, and platinum. Compared to X-ray photons, electrons are more suitable for smaller samples, such as proteins or thin 2D crystals, through which X-ray photons would pass unnoticed. Charged electrons interact more strongly with atomic nuclei, but they cannot penetrate materials thicker than a micrometer. Following electrons, interference experiments were performed illustrating the wave nature of neutrons, other quantum particles, and even atoms and molecules. Marcus Arndt’s group in Vienna has a long-term goal of observing interference with viruses.

The wave nature of matter has been experimentally verified for increasingly larger systems – many other experiments are omitted in the figure. The group of Markus Arndt in Vienna has a long-term goal of observing interference with viruses.
Author of the article: Mário Ziman, Institute of Physics, Slovak Academy of Sciences, Bratislava
Illustrations: Diana Cencer Garafová, QUTE.sk – Slovak National Center for Quantum Technologies
Translation: Gabriela Kotúčová
Image source: wikipedia public domain, www.nobelprize.org



