Spin Is In
And yet it moves! It isn’t completely clear whether Galileo Galilei ever actually uttered this sentence, which has become a symbol of facts defying dogma. But what does rotation mean for quantum systems?
Heisenberg’s uncertainty in position and momentum doesn’t allow us to talk about trajectories, not even rotational ones. We cannot measure how electrons in atoms orbit around the nucleus. Despite this we still speak of rotation in quantum systems. In physics, rotational motion is linked to angular momentum – the (vector) product of position and momentum. But it’s difficult to multiply quantities that are themselves uncertain. In the quantum world, we don’t claim that the system rotates, instead, we can determine its angular momentum partially. Thanks to this, we can discuss the consequences and properties that, for classical systems, go hand in hand with rotation – for example, the spinning of electrons in atoms. It even turns out that quantum systems are almost obsessed with rotation in this sense. They possess an inner world in which they spin, even though they don’t physically rotate. How did we discover this?
Zeeman Effect Experiment
Faraday once attempted, unsuccessfully, to measure the influence of a strong magnetic field on the light emitted by atoms. 30 years later, P. Zeeman repeated Faraday’s experiments, but used a new invention – artificially produced optical diffraction gratings. Thanks to their higher sensitivity, he observed in 1896 a splitting of spectral lines – a change in the frequencies of photons emitted by sodium atoms placed in a magnetic field. Not only did the frequencies shift with the strength of the magnetic field, but the very presence of the field also changed the number of observed lines.
Spectral lines are related to the solutions of the Schrödinger equation, which tells us what energy values are allowed for an atom. For each of these energy values there are different “realizations,” distinguished by their angular momentum. A magnetic field changes the atom’s energy structure, causing the energies to depend on angular momentum as well. The result is a new set of energy differences and, consequently, new frequencies of emitted or absorbed photons.
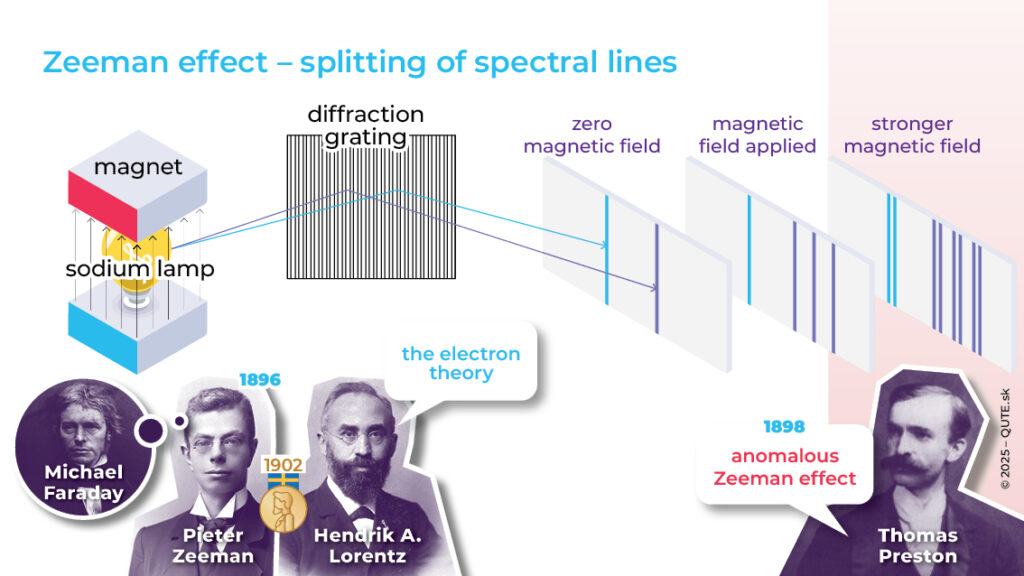
Zeeman was a student of H. Lorentz, who described the force (named after him) by which a magnetic field acts on a moving electric charge. Lorentz worked on developing a theory of electric charges and currents, which are produced by electrically charged particles (electrons were still only a hypothesis at the time), and he was able to explain the splitting of spectral lines. But that wasn’t all. In 1898, T. Preston discovered an additional splitting, even for energies corresponding to zero angular momentum. To explain the observed anomalous Zeeman effect, in which each line splits into two more, physics needed a new idea.
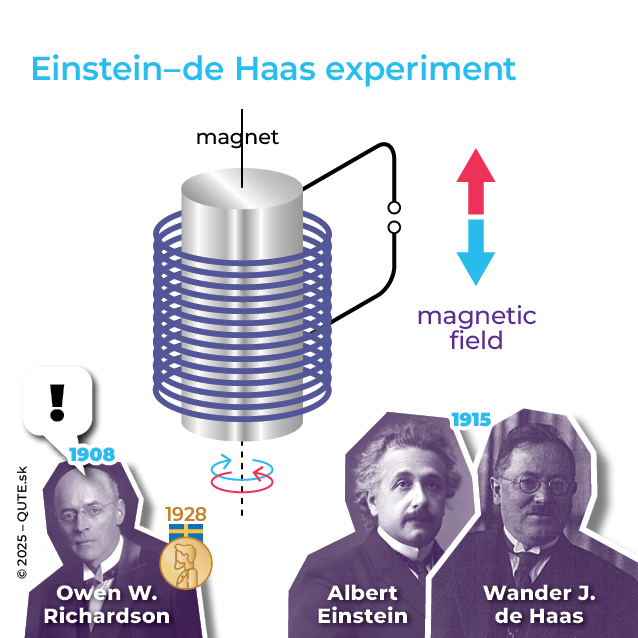
Einstein–de Haas experiment
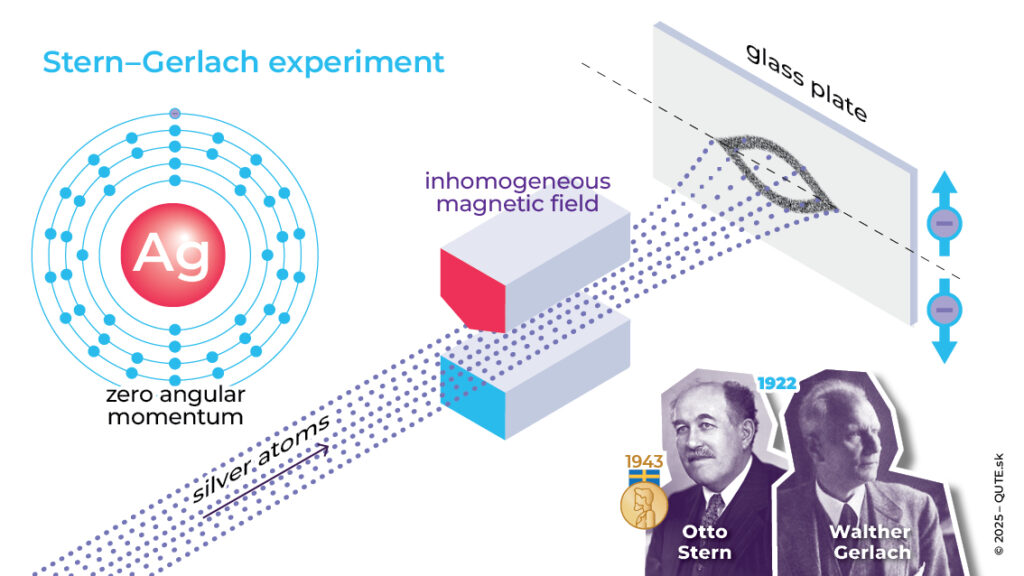
In 1921, O. Stern devised an experiment that he later carried out together with W. Gerlach. They directed a beam of silver atoms through an inhomogeneous magnetic field and observed that the beam split into two. Some atoms were deflected upward, others downward. Although an atom is electrically neutral, it can still be sensitive to a magnetic field thanks to its angular momentum. In some respects, the situation is similar to the splitting of spectral lines. However, there was a catch. Angular momentum is a three-dimensional vector, but its three mutually perpendicular components are quantum indeterminate.
We cannot measure angular momentum as a full vector. However, we can determine its total magnitude and the value of one of its components. Both of these values are quantized. The magnitude of angular momentum is limited to the values ħ l(l+1) for l= 0, 1, 2, …and when measuring its components we obtain only the values 0, ±ħ, … ±lħ . The value of the angular-momentum component in the direction of the magnetic field is precisely what determines how the atomic beam splits. This component can take on 2l+1 different values, which is an odd number. However, Stern and Gerlach observed only two.
Stern–Gerlach experiment
Quantum physics has taught us that not only radiation but also matter is quantized and cannot be divided indefinitely. Atoms, however, are not indivisible. They are made up of electrons, protons, and neutrons. Today we understand that electrons cannot be divided further, but protons and neutrons are composed of quarks. In addition to the periodic table of elements, we also have a table of elementary particles, but our understanding of what is made of what has become more complicated. It seems logical to say that if X decays into A and B, then X is composed of A and B. But the quantum world doesn’t fully follow such logic at the level of particles. There exist crystals in which an ultraviolet photon splits into two red photons, but this doesn’t mean that an ultraviolet photon is made up of two red ones. It only means that the energy of the two red photons equals the energy of the ultraviolet photon. We might say that energy not only changes form but also “dresses itself” in different particles of being. For the particles themselves, transformations (creation and annihilation) take place. For example, a free neutron undergoes beta decay, transforming into a proton, an electron, and an antineutrino.
Quantization of the world, originally motivated by the idea of dividing matter, adopted the concept of particles as the fundamental building blocks of our universe. However, we observe quantization in all physical quantities. In practice, this means that in different systems certain quantities can take on only specific values. These values are not always integer multiples of some basic quantum of the given quantity. For example, the allowed energy values of the hydrogen atom are 1/n2 multiples (for n = 1, 2, 3, …) of -13.6 eV, and the emitted photons have frequencies proportional to the differences between these energies. The quantization of atomic energy, encoded in the Schrödinger equation, solved the problem of the equations of classical physics.
Three Nuts for Two
The anomaly of the Zeeman effect lies in the fact that each spectral line splits into two; the Einstein–de Haas experiment shows that the expected proportionality constant is twice the observed one, and finally, in the Stern–Gerlach experiment, the beam of atoms splits into only two components for reasons that were initially unknown. The common denominator of all three puzzles is the strangely quantized angular momentum of the atom in a magnetic field, and the number two.
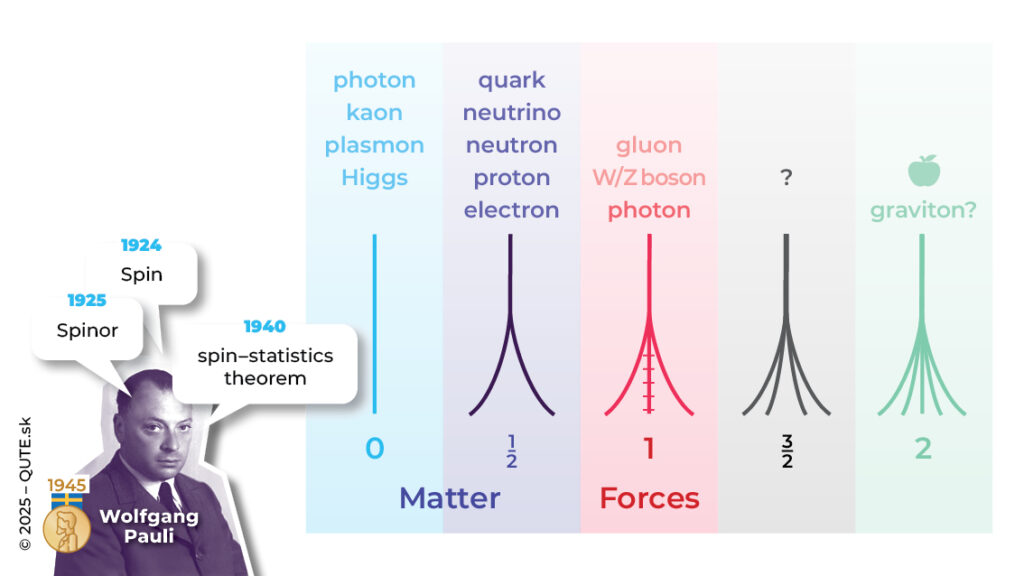
The attempt to crack these puzzles began with W. Pauli in 1924, when he introduced the “classically non-describable two-valuedness” of the electron. He rejected R. Kronig’s idea that it was actually an angular momentum associated with the rotation of the electron itself, because the required rotation speed would exceed the speed of light by several orders. The same idea was published in 1925 by G. Uhlenbeck and S. Goudsmit. Although they faced criticism, their hypothesis was used a year later by L. Thomas, who successfully explained the so-called fine structure in atomic spectra – the relativistic effect of splitting the hydrogen atom’s spectral lines in the absence of an external magnetic field.
He convinced most physicists, including Pauli, of the existence of an intrinsic angular momentum of the electron, which they called spin. “And yet it moves,” doesn’t happen here. The idea of the electron literally rotating is neither correct nor necessary. We understand spin as a fundamental property of the electron, similar to its mass or charge.It behaves exactly like angular momentum, whose component has only two possible values, ±ħ/2, the missing “two” that appears in all the experiments mentioned above.
Half Worlds
Spin is not a specialty of the electron. Every quantum system, in addition to its “orbital” angular momentum, also has its own internal rotational world – spin. Its magnitude is ħ s(s+1), but unlike orbital angular momentum, the values of spin are s = 0, 1⁄2, 1, 3/2, 2, … the so-called half-integer numbers. Electrons, protons, neutrons, quarks, and neutrinos are all particles with half-integer spin. In 1927, Pauli came up with a way to describe systems with spin within quantum physics. In this framework, the electron is described by a pair of quantum waves – one for each value of its spin component, and the pair as a whole behaves according to the Schrödinger equation.
Fermions a bosons
An interesting case is that of the photon, which has spin one. Because its mass is zero, the spin component cannot take the value zero. The values ±ħ correspond to right-handed and left-handed circular polarization. Just like the electron, the photon’s spin (its polarization) forms an intrinsically two-valued world.
Systems with half-integer spin are called fermions, and systems with integer spin are called bosons. Fermions, when they form a single system, behave “antisocially” and it’s impossible for them to share the same set of parameters (Pauli exclusion principle). Bosons, on the other hand, tend to “gather in communities,” occupying states with identical characteristics. The “antisocial” nature of electrons is crucial for the stability of matter, the periodicity of atomic properties, the conductive behavior of materials, and more. The Einstein–de Haas experiment shows that the magnetism of materials originates in the spin of the electron. The spin of nuclei is observed in NMR examinations.
Author of the article: Mário Ziman, Institute of Physics, Slovak Academy of Sciences, Bratislava
Illustrations: Diana Cencer Garafová, QUTE.sk – Slovak National Center for Quantum Technologies
Translation: Gabriela Kotúčová
Image source: wikipedia public domain



