The absorption of a photon depends on the velocity of the particle.
Quantum World
Winter Is Coming – Stop, Stop!
Temperature is a manifestation of the disordered motion of particles. The lower it is, the less the particles move. Can they ever come to a complete stop? And do we even want that?
At the beginning of the 18th century, the French physicist Guillaume Amontons discovered that, at a fixed gas volume, both pressure and temperature decrease proportionally. Based on this observation, he was the first to propose the idea that there is a lowest possible air temperature, which would occur at zero pressure. He estimated it at −240 °C, which is not far from its actual value – absolute zero, which is −273.15 °C.
Absolute zero is often imagined as the complete halt of the molecular and atomic world. However, this idea conflicts with quantum physics. Specifically, quantum uncertainty in position and velocity prevents particles from truly freezing. Quantum systems remain in motion even at absolute zero.
Zero Is Not Boring
Cooling a system means taking energy out of it. At absolute zero, there is no more energy to take away. By definition, the system has its lowest possible energy and therefore cannot lose any more. A quantum system “freezes” in its so-called ground state, yet its other properties – positions, velocities, etc. – do not take precise (zero) values. We cannot say that a quantum system at zero temperature is completely motionless.
Thanks to quantum effects, absolute zero is far from boring. On the contrary, phenomena such as superconductivity, superfluidity, Bose–Einstein condensation, and supersolidity appear – all peculiar behaviors we discover only at extremely low temperatures and through quantum physics. Many other system properties – chemical, magnetic, electrical, optical – can also be explained by knowing the ground-state energy and quantum nature of the system.
Temperature is closely tied to a system’s quantumness. At higher temperatures, systems exist in less controllable environments. We talk about thermal noise, which suppresses quantum interference effects, so quantum phenomena are rarely observable at everyday temperatures. Conversely, very low temperatures ensure that quantum properties are present and detectable in the system.
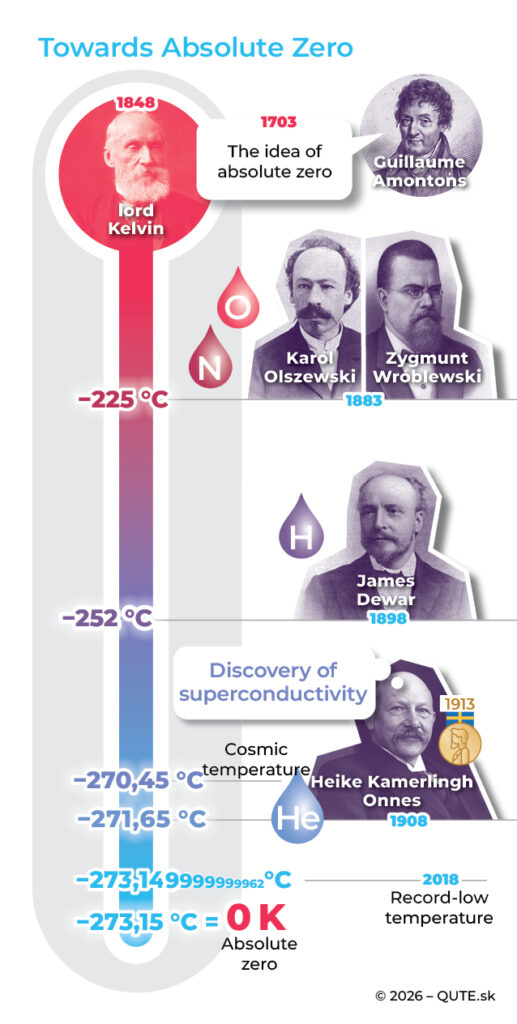
Lord Kelvin was the first to determine the value of absolute zero. The image illustrates the historical progression from the idea of absolute zero to the lowest temperature achieved to date.
Towards Zero
The exact value of absolute zero was experimentally determined in 1848 by the Scottish physicist Lord Kelvin of Largs (born William Thomson). The third law of thermodynamics tells us that, in practice, we cannot reach this temperature. But just how close can we get to zero?
Michael Faraday successfully liquefied common gases, reaching temperatures of −130 °C. He believed that some of them – like oxygen, nitrogen, and hydrogen – could never be liquefied. He was mistaken. In 1883, Zygmunt Wróblewski and Karol Olszewski at Jagiellonian University in Krakow were the first to liquefy nitrogen and oxygen, setting a low-temperature record of −225 °C. In 1898, James Dewar in London reached −252 °C and successfully liquefied hydrogen.
Liquefying helium came ten years later, thanks to the Dutch physicist Heike K. Onnes at Leiden University. Moreover, at the temperature of liquid helium (−269 °C), he observed the disappearance of electrical resistance in copper, discovering superconductivity. He reached −271.65 °C, becoming the first to achieve a temperature lower than that of interstellar space, which is about −270.45 °C.
So far, the closest anyone has come to absolute zero is researchers in Bremen, Germany. In a 2018 experiment with a Bose–Einstein condensate of rubidium atoms, they reached an astonishing −273.149 999 999 962 °C – just 38 trillionths of a degree above absolute zero. This achievement is recorded in the Guinness World Records.
Cooling with Light
The most basic method of cooling is simply bringing a system into contact with a colder object. But very quickly, we run into a problem: how do we create ever colder objects and maintain their low temperature? Over time, humans have invented a variety of sophisticated techniques to reach ever lower temperatures – including using light itself.
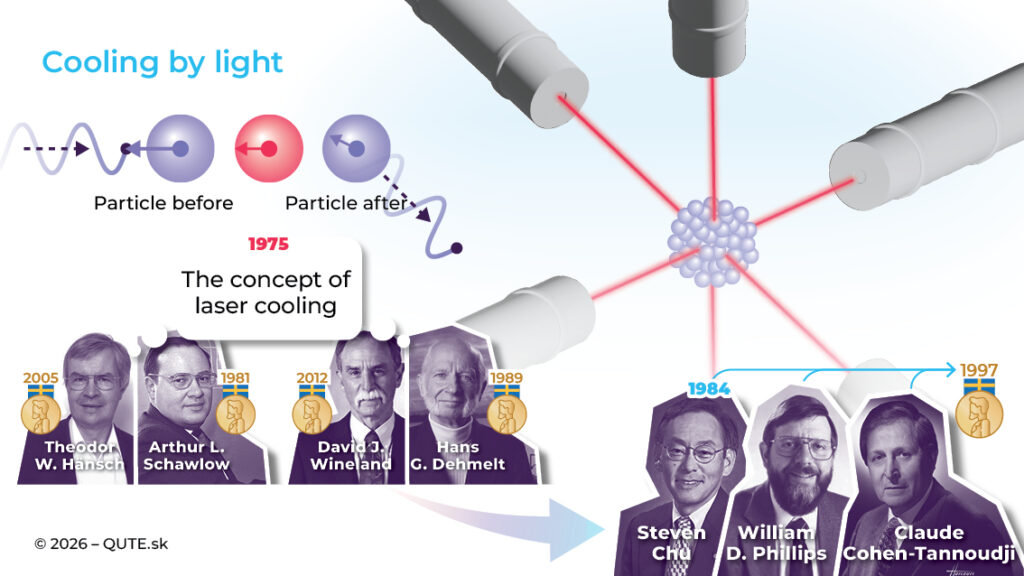
In 1975, Theodor W. Hänsch and Arthur L. Schawlow at Stanford, and independently David J. Wineland and Hans G. Dehmelt in Seattle, came up with the idea of laser cooling. In 1984, Steven Chu realized the concept and successfully cooled sodium atoms. Around the same time, William D. Phillips achieved temperatures even lower than theoretical predictions using laser cooling, which was later explained and confirmed by Claude Cohen-Tannoudji. All of them were awarded Nobel Prizes for their work.
This cooling method is based on the Doppler effect and the quantum interaction between photons of light and gas atoms. The energy and momentum of a photon are absorbed by an atom only if the photon has the right frequency corresponding to the energy difference between atomic states. The Doppler effect ensures that this frequency selectively interacts with the faster-moving atoms.
The Principle of Doppler Cooling
The Doppler effect tells us that the frequency of a wave depends on the relative speed (both magnitude and direction) of the source and the observer. That’s why, for example, the sound of a siren suddenly changes pitch as an ambulance drives past. In laser cooling, the laser is tuned to a frequency slightly lower than the one the atom naturally absorbs. A stationary or slow-moving atom will barely notice these photons, but faster atoms moving toward the light source will interact with them.
When a photon is absorbed and then re-emitted, the atom’s momentum changes as well. Imagine an atom moving to the right against an incoming laser. When it absorbs a photon, it takes on the photon’s opposite momentum and slows down slightly. Upon re-emission, it regains momentum in the opposite direction of the emitted photon. Since the emission direction is random, the atom’s momentum to the right is, on average, reduced. The result is a net slowing of the atom in that direction.
To slow atoms in all directions, the same principle must be applied simultaneously along every axis. This is done by shining lasers from all six sides. Thanks to the Doppler effect, the faster atoms absorb the laser photons and re-emit them randomly. On average, their speed decreases, and they essentially “freeze” at the intersection of all the lasers.
Particles in a Trap
To keep cooled particles cold, scientists use devices called traps, which generate force fields that hold the particles “frozen” in space even after the cooling process. For atoms, magneto-optical traps are used. For charged particles, ion traps are employed, which confine ions using electromagnetic fields. These traps themselves are maintained in environments around −265 °C.

The image shows charged grains of flour trapped in a confinement, behaving similarly to ions; unlike ions, however, they are visible to the naked eye.
In 1953, Wolfgang Paul and Helmut Steinwedel designed and patented the Paul trap, which confines particles using purely electric fields. In 1959, Hans G. Dehmelt invented another type of trap combining magnetic and electric fields, named the Penning trap after the Dutch physicist Frans M. Penning, whose experiments inspired its design. Penning traps are used, for example, in experiments with antimatter.

NIST quantum chip processor in which two beryllium ions, separated by 40 micrometres, operate as qubits.
From Absolute Zero to Logical Zero
Using lasers, we can cool particles to just a thousandth of a degree above absolute zero and store them in traps, where they can be individually manipulated – essentially like tiny computing elements. It is therefore no surprise that the first realizations of quantum computing used particles in traps.
In 1995, Ignacio Cirac and Peter Zoller from Innsbruck proposed a concrete design for quantum computing using cold ions in a linear Paul trap. Each ion represented a single quantum bit (qubit), and computations were carried out using laser pulses. That same year, a two-qubit version using beryllium ions was realized at NIST in the United States under the leadership of D. J. Wineland.
In November 2025, Quantinuum unveiled the breakthrough 98-qubit Helios quantum processor, operating with barium ions. Helios is currently the most precise and powerful quantum processor. This “computing trap” provides up to 48 logical qubits and operates at −258.15 °C. For more than a century, low-temperature physics has been full of useful and surprising quantum phenomena.
Author of the article: Mário Ziman, Institute of Physics, Slovak Academy of Sciences, Bratislava
Illustrations: Diana Cencer Garafová, QUTE.sk – Slovak National Center for Quantum Technologies
Image source: wikipedia public domain (Y. Colombre, NIST)



