Through Barriers Without Obstacles
- Home
- Quniverse
- Quantum World
- Through Barriers Without Obstacles
Through Barriers Without Obstacles
To overcome obstacles, we need energy. If, for any reason, we lack that energy, our only options are to bypass the obstacle, remove it, or accept it. A quantum system, however, has a different experience despite lacking the necessary energy, it still has a chance to overcome the obstacle.
The word energy comes from Greek and expresses the measure of a physical system’s ‘activity.’ However, the uncertainty and randomness of quantum physics allow us to assign a definite value of energy to a system only in special cases. What we can talk about, instead, are the probabilities of energy values, and by solving the Schrödinger equation we can predict which energies are actually possible.
Alone in a Well
Let us consider a situation in which a quantum system is confined to a one-dimensional space with infinitely high and thick walls. We can also picture this as an infinitely deep well. Mathematically, this means that the entire world of the system is a line segment on which it is trapped. Quantum physics tells us that the probability of an energy value being nonzero only exists if it’s a so-called “square” multiple (1, 4, 9, 16, 25, …) of e energy E = h2/8mL2, where h is Planck’s constant, m is the mass of the system, and L is the width of the space. The greater the mass and the width of the space, the denser the allowed energy values, but the energy itself is not upper bound.
Infinite walls or wells are, of course, a physicist’s idealization. In the more realistic case of finite parameters, the allowed energy values become non-integer multiples of the same base value E. Intuitively, the height of the wall or the depth of the well determines the energy required for the system to escape beyond the confined space. Yet, surprisingly, for quantum waves of all allowed energies, even the smallest ones, there exists a nonzero probability that the system is also found outside. If we make the wall thin enough, then along with the system we discover that it has a chance to appear beyond the wall and continue moving. How did it get there? In a mysterious process, it “tunneled through.”
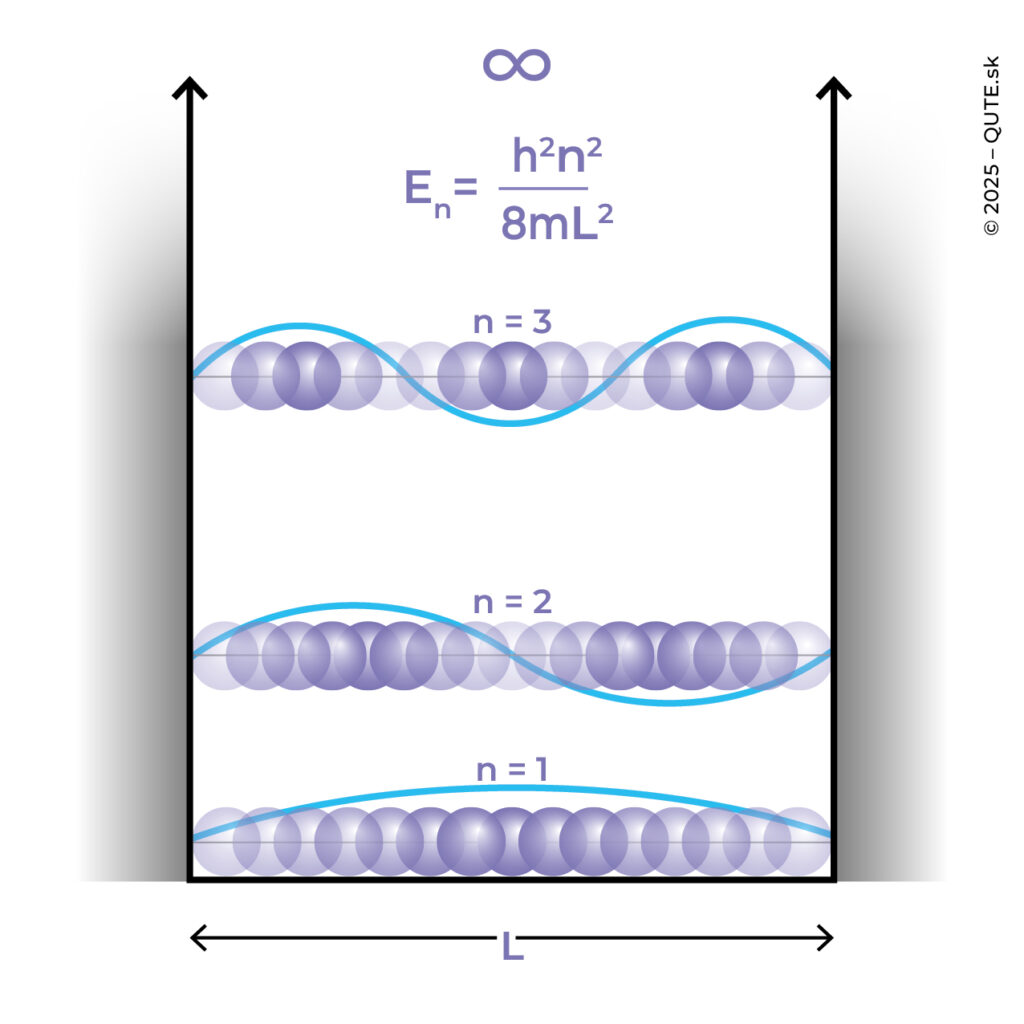
System Confined in an Infinite Well
Tunneling Without a Tunnel
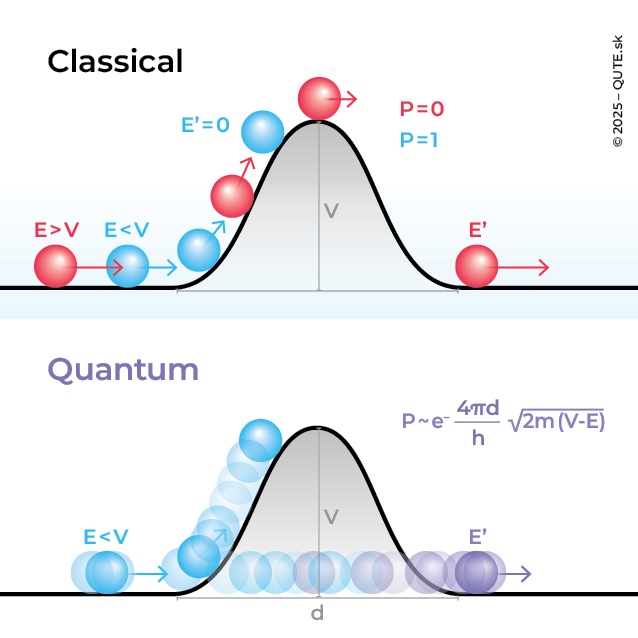
Quantum tunneling
Quantum tunneling is an important and extremely useful manifestation of the principles of the quantum world. The phenomenon was first discovered by Friedrich Hund in 1927, when he studied solutions of the Schrödinger equation for diatomic molecules, where an electron exhibited tunneling between atoms. That same year, Lothar Nordheim came up with a textbook example of tunneling – the passage of a quantum system through a potential barrier with a rectangular profile (walls). The probability that a particle will tunnel through a barrier depends on its mass, its energy, and the length of the barrier. The heavier the object, the less distance it can penetrate into the barrier. Its ability to tunnel decreases exponentially with its mass. For this reason, tunneling is most commonly observed in electrons, but there are also important examples involving heavier objects. In 1932, John Uhlenbeck studied the ammonia molecule NH3 and described the tunneling of the nitrogen atom through the plane formed by the three hydrogens.
Nuclear Tunnels
In 1928, George Gamow came up with the idea of explaining the alpha radiation of uranium through quantum tunneling. At that time, little was known about the structure and functioning of atomic nuclei. It was understood that alpha radiation consisted of positively charged particles, unsurprisingly named alpha particles. Today, we know that an alpha particle is the nucleus of a helium atom, composed of two protons and two neutrons. It was assumed that alpha particles, like other nucleons – the particles that make up the nucleus, are held together by what were at the time hypothetical nuclear forces, which compete with the electrostatic repulsion of the positive charges within the nucleus. G.Gamow assumed that the emission of an alpha particle occurs at the moment when the particle, trapped inside the nucleus by nuclear forces, tunnels beyond the sphere of their influence. His calculations explained why nuclear decay is exponential and also why short-lived isotopes emit more energetic alpha particles than long-lived ones. This naturally raises the question: why don’t atomic nuclei emit clusters of nucleons other than helium nuclei? The answer is that they do. The helium nucleus is among the most stable configurations, meaning that protons and neutrons tend to cluster into alpha quartets. Tunneling of potentially even more strongly bound nucleons is limited by their higher mass and lack of energy to cross the nuclear force barrier. Nevertheless, emissions of carbon nuclei 14C have been observed in the decay of francium 221Fr and radium 222Ra, oxygen 20O in the decay of thorium 228Th, and others. These are relatively rare processes, but they do exist. Similarly, emissions of individual protons and neutrons have also been observed.
Conductive Tunnels
Fascinating physics occurs at the interfaces of two different materials, where two distinct, both quantum, worlds literally meet. Depending on the charge carriers (negatively charged electrons or positively charged holes), we distinguish between two types of semiconductors. By “gluing” them together, we obtain an electronic component that conducts current only in one direction – a so-called rectifier, or diode. There is a threshold voltage in the forward direction at which the diode begins to conduct current. The phenomenon is rooted in quantum physics (a story for another time), but not in tunneling
In 1957, Japanese physicist Leo Esaki created the tunnel diode, in which electrons tunnel through the barrier between semiconductors, enabling current flow even at sub-threshold forward voltages. Another remarkable effect occurs when, as the voltage increases, the current actually decreases (the exact opposite of Ohm’s law), until a critical voltage is reached, after which the diode “switches on” again in the standard regime. As the voltage rises, the parameters required for electron tunneling change: specifically, the free holes in one semiconductor and the free electrons in the other semiconductor no longer have similar energies. This raises the energy barrier and reduces the chance that the holes will be filled by tunneled electrons.
In 1960, Ivar Giaever experimentally studied electron tunneling through a thin insulating layer between aluminum and lead, depending on whether they were in a superconducting state. He demonstrated the existence of the so-called energy gap in superconductors, thereby confirming a key prediction of the superconductivity theory. Soon after, Brian Josephso, still as a student, theoretically showed that not only electrons but also superconducting Cooper pairs can tunnel through a thin insulating layer, even in the absence of an applied voltage. This led to the creation of another electronic component that is widely used today (a superconductor–insulator–superconductor), which bears his name: the Josephson junction.
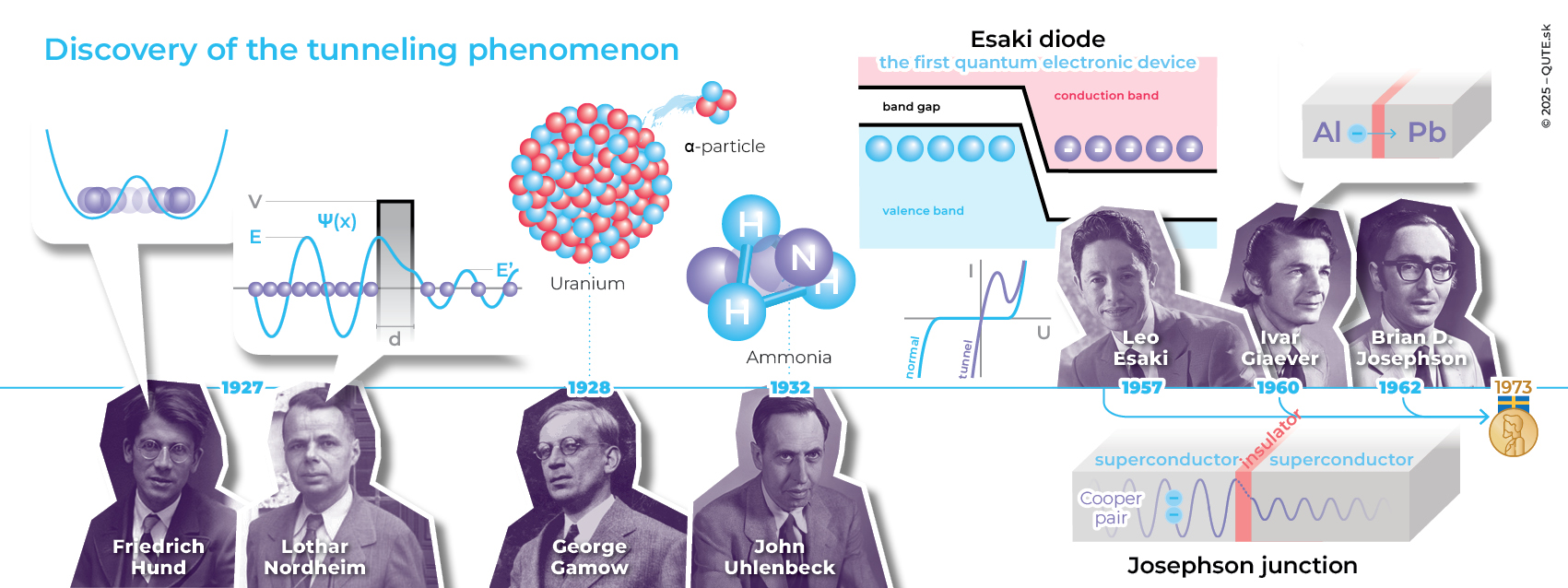
The story of quantum tunneling
A View Through the Tunnel
Our electronics are full of electron quantum tunneling. But the applications of tunneling don’t stop there. It also plays a crucial role in microscopy. In 1981, Gerd Binnig and Heinrich Rohrer invented the scanning tunneling microscope (STM). The STM consists of a tip just one atom sharp, which passes within less than 1 nm of a metallic surface and records the electrons that have tunneled from the material. The STM operates in two modes: it either maintains a constant voltage and height above the surface, or it keeps a constant current while piezoelectric electrodes adjust the tip’s position by changing the voltage. Beyond surface mapping, the STM can also manipulate individual atoms, enabling the creation of new nanostructures and nanomaterials.
Finally, it is worth mentioning that tunneling has a lesser-known counterpart. Systems with insufficient energy to “jump over” a barrier naturally reflect off it. However, knowing about quantum tunneling, we shouldn´t be surprised that reflection can also occur for systems that would seemingly have enough energy to “jump over” the barrier. Instead of opening a fictitious quantum tunnel, we can think of it as erecting a fictitious quantum wall, which with a certain probability prevents the system from overcoming the real obstacle.
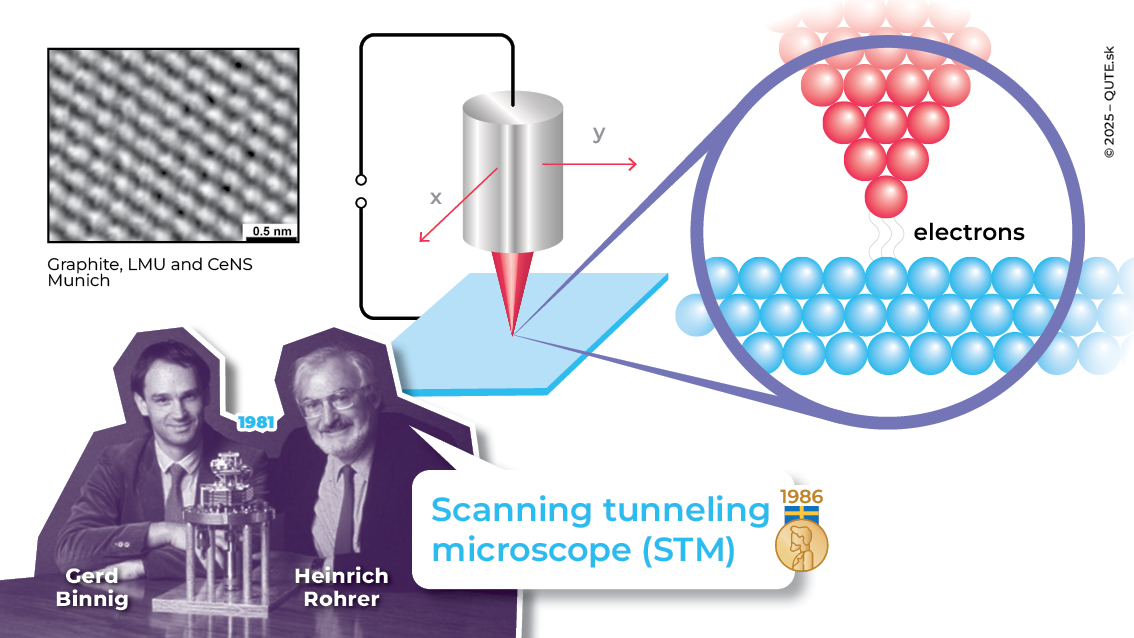
Scanning tunneling microscope
Author of the article: Mário Ziman, Institute of Physics, Slovak Academy of Sciences, Bratislava
Illustrations: Diana Cencer Garafová, QUTE.sk – Slovak National Center for Quantum Technologies
Translation: Gabriela Kotúčová
Image source: wikipedia public domain, www.nobelprize.org, arxiv.org/pdf/1403.0059, Image courtesy of IBM Research Zurich



