Photoelectric effect. The image shows that if we split light into its individual color components, photons of certain colors will not release any electrons from the material. For higher-energy photons, electrons are emitted from the material with different velocities, which depend only on their frequency (color).
QUANTUM WORLD
The Invisible Photon
Temperature, among other things, determines the amount of heat that objects emit in the form of electromagnetic radiation. The human body “glows” in the infrared, yet its radiation is invisible to us without a thermal camera. When heating iron, however, we can see that different temperatures correspond to different colors.
Thermal Radiation
Physicists in the 19th century studied the relationship between radiation and temperature, yet experimental results were in a fundamental conflict with the existing physics. Calculations predicted an infinite amount of ultraviolet radiation, but in reality this radiation is minimal. In fact, thermal radiation is dominated by the infrared component, which we associate with the sensation of warmth, for example, when basking in the sun’s rays.

Superconducting nanowire single-photon detector
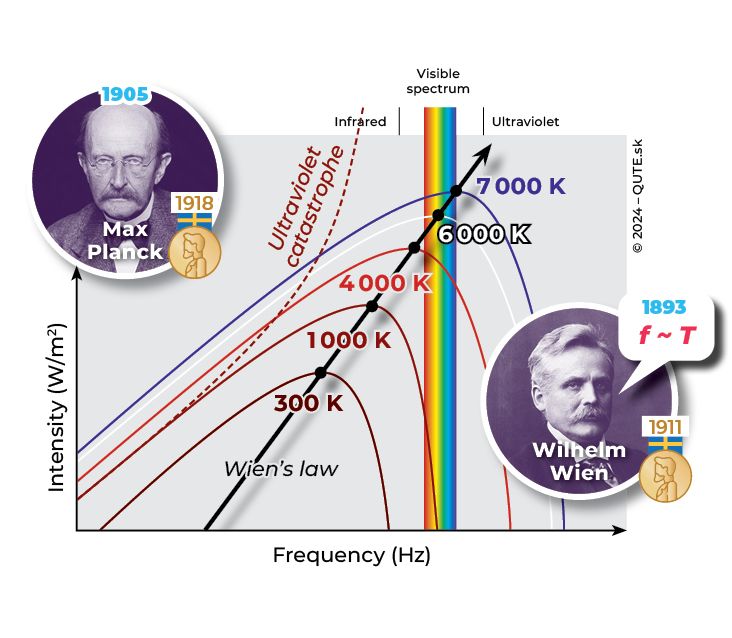
Black-body radiation. Late 19th-century physics predicted a catastrophic scenario for the dependence of radiation intensity on its frequency (the so-called ultraviolet catastrophe, shown as a dotted line). Using the concept of the photon, Max Planck explained the observed radiation intensities for different temperatures. For example, at 6 000 K (the temperature of the Sun’s surface), all colors in the visible frequency range have approximately the same intensity. This is why we observe its radiation as white light.
In 1900, Max Planck attempted calculations based on the assumption that the energy of the radiation under study could only be an integer multiple of a certain fundamental quantum of energy. According to M. Planck, the reason lies not in the radiation itself, but in the oscillations of atoms in the material, which create standing waves (as on a guitar) and therefore cannot have arbitrary frequencies and energies to be transferred in the form of radiation. He proposed that the size of an emitted quantum of energy is directly proportional to the frequency of the radiation. This means that an ultraviolet quantum carries more energy than a visible or an infrared one. With this clever approach, M. Planck succeeded. He achieved an uncannily accurate match with observations and was able to determine the proportionality constant between a quantum’s energy and its frequency. Today, it is known as Planck’s constant and has a value of h = 6,626 070 15 × 10-34 J · s-1 . M. Planck himself, however, did not associate energy quanta with a particle-like nature of radiation, but rather with the properties of matter. It was only further experiments and their interpretations that convinced him of the fundamental existence of quanta of electromagnetic radiation.
Photoelectric Effect
Imagine supplying the atoms in a material with energy that causes the electrons within them to increase their kinetic energy. Intuitively, one would expect that there is a certain critical amount of supplied energy at which the electrons are released from the atoms of the material. It came as a surprise to physicists when they discovered that this is not entirely the case if the energy is delivered in the form of electromagnetic radiation.
In 1887, Heinrich Hertz observed in his experiments that ultraviolet radiation made the sparking between two electrodes stronger, even at lower light intensities. He discovered that, unlike visible light, ultraviolet radiation released charges – electrons – from metals much more efficiently. The number of electrons emitted as a result of incident radiation is not determined solely by its intensity. If we were to shine visible or infrared light of any intensity on zinc, copper, or iron, no electrons would be emitted. However, if we used ultraviolet radiation, electrons would be released, and their number would depend on the intensity of the radiation. In that case, we would observe the photoelectric effect.
It turned out that, in order to release an electron using electromagnetic radiation, the radiation must have a certain critical frequency (its value depends on the material). Any radiation with a lower frequency has no chance, while any radiation with a higher frequency will release electrons. Another important observation, which contradicts our everyday experience, is that radiation with a supercritical frequency will release an electron even at minimal intensity, in other words, there is no need to wait for the electron to “collect” the energy. But that is not all.
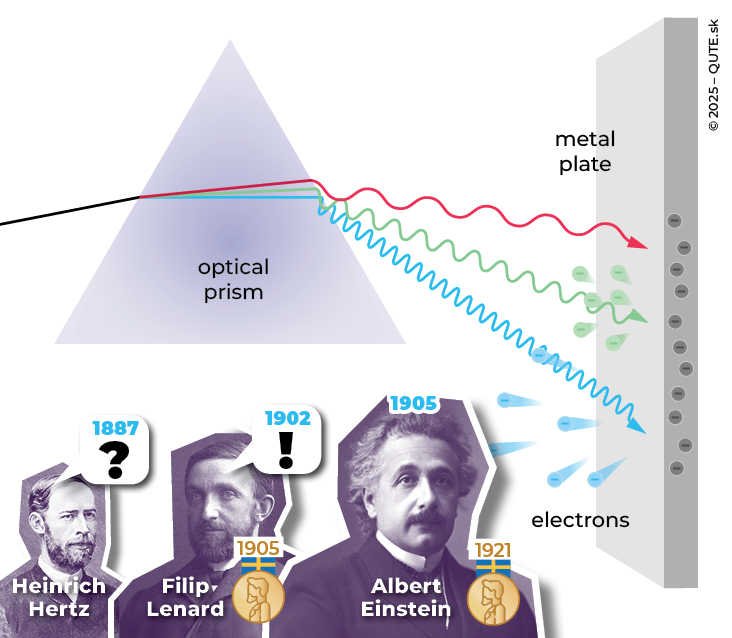
Another peculiarity of the photoelectric effect was uncovered by Filip Lenard, a native of Pressburg (present-day Bratislava) of Hungarian origin, who at the time of the discovery of the photoelectric effect was working as an assistant to H. Hertz. F. Lenard became an expert in cathode-ray tubes, in which he systematically studied the properties of the photoelectric effect. In 1902, he discovered that individual electrons released in the photoelectric effect all have exactly the same kinetic energy, and that this energy depends not on the intensity of the incident radiation but only on its frequency. How is that possible?
Setting the Stage for the Particle of Light
In 1905, everything was ready for Albert Einstein to put forward the idea that electromagnetic radiation itself is composed of indivisible quanta of energy — particles that, in 1926, the chemist Gilbert N. Lewis named photons (particles of light). The intensity of the radiation represents the number of photons it contains. The greater the intensity, the more photons there are. The frequency of the radiation determines the energy of a single photon according to Planck’s relation E = h · f .
With the concept of the photon explaining the peculiarities of the photoelectric effect becomes quite straightforward, without the need to involve the complexities of quantum physics itself, which in fact began to take shape at this very moment. Electromagnetic radiation is a stream of photons that penetrate the material and collide with electrons. An electron requires a certain critical escape energy to break free from the atom. If a photon does not possess this energy, the electron cannot acquire it. If it does, the electron is released, and its kinetic energy is equal to the difference between the photon’s energy and the electron’s escape energy. In other words, each released electron has the same kinetic energy, exactly as measured by F. Lenard. The total number of released electrons is related to the total number of photons in the incident radiation.
To See a Photon
From the total energy of electromagnetic radiation, we can determine the number of photons it contains. A typical light bulb used to illuminate a room produces on the order of 1020 photons every second. About 108 photons enter our eye each second, yet our receptors are so sensitive that they can register even a single photon. However, in an average person, the brain typically receives a signal only if at least five photons arrive within a tenth of a second, which is roughly the period at which the brain processes new information.
Detecting a single photon on the retina is perhaps not so surprising, since at the atomic scale a single photon can affect molecules and atoms. The situation becomes more complicated, however, if only one photon is truly directed into the material. We now know that a large fraction of a material’s volume is “empty” space, and that electromagnetic radiation in a material is not only absorbed but also reflected or transmitted. All of this reduces the chances of the photon making its presence known. The key to detecting a photon lies in the visible physical changes that a single photon can trigger. A typical example is so-called phase transitions, in which a small change in external parameters can completely change the nature of an object, for instance, turning water into ice.
Revolutionary Detectors
The development of single-photon detectors is a highly active area of theoretical and engineering research. The current leaders in single-photon detection are superconducting nanowire single-photon detectors (SNSPDs). Their detection principle is based on heating the nanowire by an absorbed photon, which causes it to lose its superconductivity, and the resulting sharp increase in resistance is interpreted as the detection of a photon. For photons at the standard telecommunication wavelength of 1 550 nm, SNSPDs have been shown experimentally to achieve a detection efficiency of over 98% and can detect a new photon every 30 nanoseconds.
A minor drawback is the low operating temperature of only a few kelvins. Building an in-house prototype of an SNSPD detector and deploying it in a quantum communication network is one of the goals of the skQCI project. Single-photon detectors have the potential to launch a revolution in many fields – for example by improving communication with distant satellites, increasing the sensitivity of medical and materials imaging methods, creating a quantum-secured communication layer, or even aiding in the study of exoplanets and dark matter.
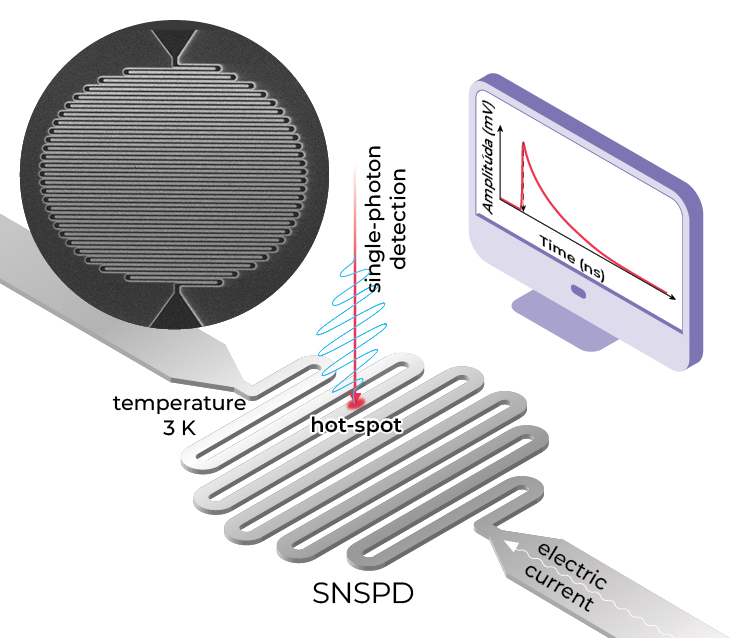
Superconducting nanowire single-photon detector
Author of the article: Mário Ziman, Institute of Physics, Slovak Academy of Sciences, Bratislava
Illustrations: Diana Cencer Garafová, QUTE.sk – Slovak National Center for Quantum Technologies
Translation: Gabriela Kotúčová
Image source: wikipedia public domain



