Exchanges Without Change
Perfectly identical twins probably don’t exist, but let’s imagine that they do. We can´t distinguish them. We cannot tell them apart based on their qualities, but each always has its own well-defined history and identity. It makes sense to give them names. Quantum particles of the same type, such as electrons, are also identical, but attributing individual identity to them is meaningless.
Quantum uncertainty has taught us that as soon as a quantum system has multiple possible ways to move, we can’t answer the question of which path it actually takes. There is no such thing as a real trajectory. If we combine this property with the fact that quantum systems of the same type are indistinguishable, we encounter another important quantum surprise.
Indistinguishability, or identity, means that in an experiment with multiple electrons, if we detect one of them, we cannot say which one it was. A system of multiple identical particles is symmetric with respect to their exchange. In practice, this means that we can give names to the particles, but if we swap the names in our description, nothing observable changes. The quantum description still gives the same predictions.
Probability Amplitudes
The omnipresent randomness of observable properties in quantum systems forces us to use the language of probability when describing the quantum world. However, quantum uncertainty implies that this language is not rich enough. We use something “more” – quantum waves, which carry probabilities in a similar way that ordinary waves carry energy.
When sound waves propagate, we talk about fluctuations in density, for waves on a lake about fluctuations in the height of the water surface, for electromagnetic waves about fluctuations in the intensity of an electromagnetic field. These fluctuations describe how the value of a physical property changes in time and space around some reference point, forming a wave that rises and falls. Such deviations are called amplitudes of density, amplitudes of surface height, or amplitudes of electromagnetic field intensity. Physics tells us that the energy carried by a wave is proportional to the magnitude (absolute value, since it can be negative) of these amplitudes. Specifically, to the square of their absolute value. The larger the amplitude, the more energy the wave carries.
In the case of quantum waves, we are not talking about transferred energy but about transferred probability P. The amplitudes of a quantum wave are called probability amplitudes and are denoted by the Greek letter ψ. These amplitudes are our description of quantum reality. Unlike surface waves, probability amplitudes are not directly observable. They encode the probabilities of all properties that are observable. The probability P(x) of finding a particle at a specific position x is given by the square of the absolute value of the current position amplitude ψ(x) at that point, written as P(x) = | ψ(x)|2.
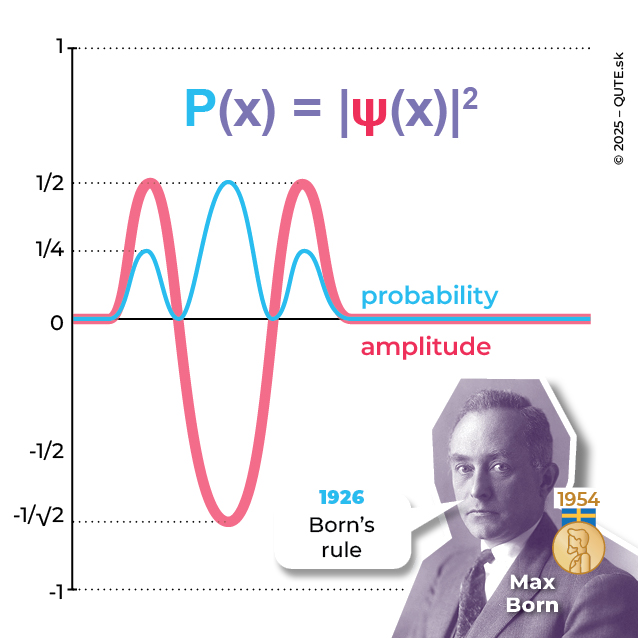
Quantum wave. The relationship between the probability amplitude (red) and the probability (blue).
Exchange Symmetry
Any change in a system’s properties means that the probability amplitudes, and therefore the quantum wave itself, must have changed. The reverse, however, isn’t always true. Since we don’t directly observe the amplitudes of quantum waves but only their absolute values, then not every change in the amplitudes leads to a change in the properties of the system. A quantum wave is a function that assigns a complex number to physical properties (for example, position), x→ψ(x), where x represents any given property. The amplitudes are complex numbers whose absolute value |ψ(x)| remains unchanged if multiplied by another complex number c that has an absolute value of one |c| = 1. In mathematics, this is expressed as |c ∙ ψ(x)|=|c|∙|ψ(x)|.

Quantum indistinguishability has two faces – bosons and fermions.
When we talk about exchanging identical particles in a way that doesn’t lead to any change in physical properties, the multiplying factor c is no longer arbitrary. If we perform the same exchange twice, we effectively do nothing. The first exchange transforms the wavefunction as ψ(x)→c ∙ ψ(x), and the second one as c ∙ ψ(x)→c2 ∙ ψ(x). The requirement that nothing has changed means ψ(x) = c2 ∙ ψ(x), and therefore c2 = 1. Even for complex numbers, this equation has only two solutions: c=±1.
In other words, the probability amplitudes of the quantum wave have the property that upon exchanging two particles, they either remain the same ψ(x)→ψ(x), or they become opposites ψ(x)→-ψ(x). We have just derived that there are only two results for particle exchange: quantum waves are either symmetric (their particles are called bosons) or antisymmetric (their particles are called fermions).
Photon symmetry
In 1924, the Indian physicist Satyendra Nath Bose published a paper revealing that the identical nature of photons affects the statistical properties of quantum waves and derived Planck’s law for blackbody radiation. We can illustrate this with the example of two coins. What is the probability of getting different outcomes when tossing two coins at the same time? Half, because there are four possibilities: heads-heads, heads-tails, tails-heads, and tails-tails, and two of these correspond to different outcomes.
While the coins are identical, they still have their own identity. Photons, however, are identical and they, due to quantum uncertainty, lose any individual identity. A photon passing through a polarizer behaves like a coin— it has a 50% chance to pass (heads) and a 50% chance to be blocked (tails). But when two identical photons meet at a polarizer, the probability that one passes (heads) and the other is blocked (tails) is not 50%, but one-third. Why? Because if we take their identical nature literally, we cannot distinguish which photon is heads and which is tails. The outcomes tails-heads and heads-tails are not two different results.
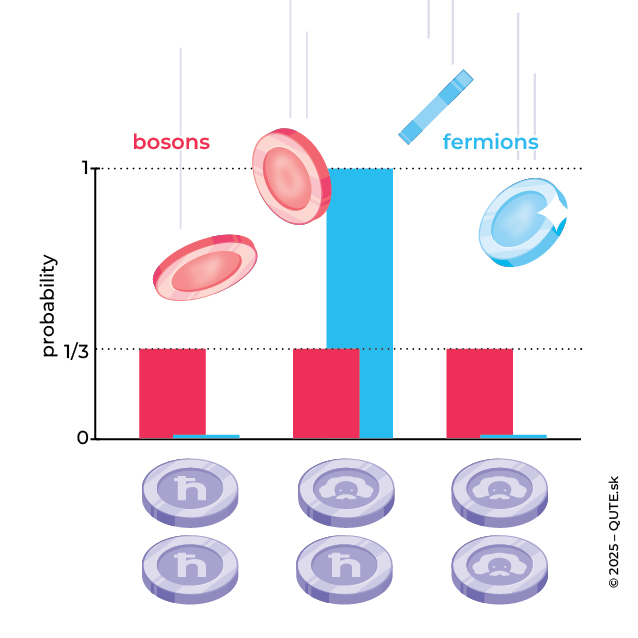
Grouping vs. exclusion. If we flip two bosonic coins at the same time, we observe three possible outcomes (Bose–Einstein statistics). Two fermion coins, however, always end up with opposite values (Fermi–Dirac statistics).
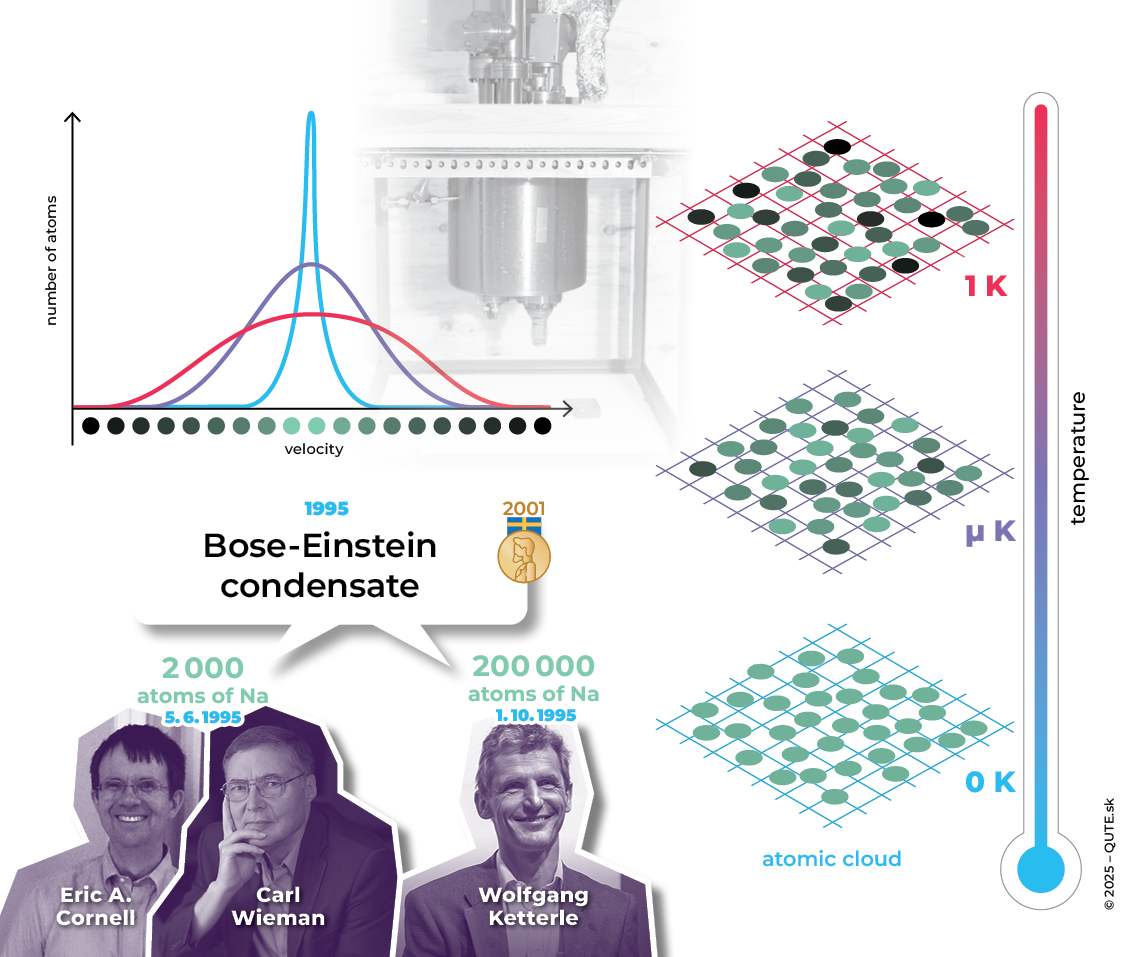
Bose–Einstein condensate – the fifth state of matter. At temperatures close to absolute zero, all bosons share the same properties. This phenomenon was first observed in 1995 in systems of Rb and Na atoms and was later awarded a Nobel Prize.
S. N. Bose was originally unsuccessful in publishing his paper. Fortunately, he sent it to A. Einstein, who recognized its significance, translated it into German, and published it on Bose’s behalf in a German journal. Einstein extended Bose’s ideas to atomic systems and predicted the existence of the fifth state of matter – the Bose–Einstein condensate.
Antisymmetry of Electrons
The quantum physics of atoms began as the physics of electrons moving with fixed energies around positively charged atomic nuclei. In a sense, the equations of quantum physics confined electrons to energy levels and prevented them from collapsing into the nucleus. However, they didn’t forbid all electrons from occupying the lowest energy level. The differing properties of various atoms suggested otherwise. In 1924, Wolfgang Pauli formulated the so-called exclusion principle, which forbids two electrons in an atom from having the same quantum numbers that describe possible quantum waves with precisely determined energies. This principle is directly related to the identical nature of electrons. Just like photons, we cannot say which electron is which, even if they have different quantum numbers. Unlike photons, the amplitude of an electron’s quantum wave changes sign when electrons are exchangedIf two electrons had the same quantum numbers, their combined quantum wave would be the product of the individual electron waves ψ(x1 ,x2 )= ψnlms (x1 ) ψnlms (x2 ), where nlms denotes the four quantum numbers (principal, angular momentum, magnetic, and spin). The exchange x1↔x2 doesn’t change the sign of ψ(x1 ,x2), which rules out the possibility of two electrons having identical quantum numbers. Electrons in an atom fill unoccupied quantum numbers and form a collective quantum wave that prevents assigning specific quantum numbers to individual electrons. Pauli’s exclusion principle is crucial for understanding many properties of atoms, including their periodicity and stability.
Bosons vs. Fermions
If quantum systems are identical, there are only two possible ways this can manifest. Bosons tend to share the same properties, whereas fermions do not allow their properties to be identical. This results in Bose–Einstein statistics for clustering bosons and Fermi–Dirac statistics for mutually exclusive fermions. The type is determined by spin— a kind of intrinsic world of the particle. Its value decides whether the system behaves as a fermion (half-integer spin) or a boson (integer spin).
Author of the article: Mário Ziman, Institute of Physics, Slovak Academy of Sciences, Bratislava
Illustrations: Diana Cencer Garafová, QUTE.sk – Slovak National Center for Quantum Technologies
Translation: Gabriela Kotúčová
Image source: wikipedia public domain, www.americanhistory.si.edu



