Between us, atoms
Chocolate bars suggest that it’s possible to share them. But what happens when only the last piece is left? If we have a knife, it’s no problem to cut it into two smaller ones. And then into even smaller ones. But does the last piece exist?

Historical Development of the Atomic Model
From Shape to Nuclear Charge
The ancient Greek philosopher Democritus imagined a world in which the last indivisible piece exists, and he named it “uncuttable, indivisible” – in Greek, átomos. He also claimed that atoms were not tightly glued together, but that the space between them was filled with void. According to his philosophy, the properties of matter were determined by the shapes of atoms – for example, the atoms of fire were sharp. At the beginning of the 19th century, the idea of atoms helped scientists understand the rules of chemical processes. In 1808, based on his observations, John Dalton formulated a theory according to which chemical substances are groupings of elementary elements, occurring in compounds in whole-number ratios. In homage to Democritus, he called them atoms. During chemical reactions, these atoms are rearranged, which changes their ratios and therefore the chemical compounds themselves.
Throughout the 19th century, different atoms were gradually identified. By analyzing chemical reactions, scientists were able to determine their relative masses. Chemists also noticed that when atoms were arranged in order of their mass, certain chemical properties of the elements repeated. In 1869, Dmitri I. Mendeleev published the Periodic table of the chemical elements. The periodicity of chemical properties suggested that the atomic world follows certain rules. It was only 57 years later that quantum physics finally helped us understand them.

The Periodic Table of Elements. The first list of elements was compiled by J. Dalton. When arranging the elements, D. I. Mendeleev predicted four new elements (now known as scandium, gallium, germanium, and technetium) to fill the gaps in the table, thereby confirming its validity.
In 1897, Joseph J. Thomson experimentally confirmed that cathode rays have mass and are composed of corpuscles that are 1800 times lighter than the lightest known atom – hydrogen. The corpuscles are electrically charged, and he correctly identified them as electrons – the particles of electric current predicted by George J. Stoney in 1874. Since cathode rays are emitted by the atoms of the cathode, electrons must therefore reside within these atoms, which means that something can indeed be “cut off” from the atom.
The question of how electrons are contained within atoms was later studied experimentally by Ernest Rutherford. Using radioactive alpha radiation in 1909, he discovered that positive charge is concentrated in a relatively small, central part of the atom. He thus discovered that the atom has a positively charged nucleus, around which the light electrons move. In 1911, lawyer and amateur physicist Antonius J. van den Broek proposed rearranging the elements in the Periodic Table according to the charge of the atomic nucleus (which E. Rutherford later called the atomic number), giving the table its modern form.
Predictions and Reality
The chemical periodicity and the properties of atoms are explained by the fundamental equation of the quantum world – Schrödinger’s equation. According to this equation, the hydrogen atom is a system consisting of a positively charged proton and a negatively charged electron of negligible mass. The attractive electric force creates a physical system similar to the gravitational force between the Sun and the Earth. However, the equations of the 19th-century physics predicted a “catastrophic” scenario for such a planetary model of the atom. An electron moving around the nucleus inevitably loses energy in the form of electromagnetic radiation. As a result, it would slow down and, within an incredible 10⁻¹¹ seconds, collapse into the nucleus. Yet nothing like this actually happens.
Niels Bohr, in his explanation of the hydrogen atom, simply forbade such radiation from the electron. He connected emission with the idea of the photon and identified that atoms emit only photons of certain frequencies, corresponding to discrete jumps in their energies. In Bohr’s model, electrons revolve around the nucleus only along certain orbits. The set of emitted frequencies is called a spectrum, and each atom has its own unique spectrum. For example, the presence of the yellow frequency 510,29 THz in the radiation from the Sun led to the discovery of a new “extraterrestrial” element ,helium, in 1868. Today, by analyzing spectra, we can identify atoms and molecules and thus determine the chemical composition of distant stars and galaxies.
Schrödinger’s equation indeed predicts that the electrons and protons of an atom have certain preferred energies, and radiation occurs only if the atom is in an excited state. Under ordinary conditions, only the electrons farthest from the nucleus become excited. When an excited electron “jumps” to a lower energy level, the atom loses energy in the form of a single emitted photon whose energy E equals the lost energy. The frequency of the emitted photon is given by Planck’s relation , where h is Planck’s constant.
Quantization of the World
Quantum physics has taught us that not only radiation but also matter is quantized, and we cannot divide it infinitely. Atoms, however, are not indivisible. They are made up of electrons, protons, and neutrons. Today, we understand that electrons cannot be divided any further, but protons and neutrons are composed of quarks. In addition to the table of chemical elements, we also have a table of elementary particles (Standard Model of Elementary Particles) – yet our understanding of what is made of what has become more complicated. It is logical to say that if X decays into A and B, then X is composed of A and B. But the quantum world does not fully share this logic at the level of particles. There exist crystals in which an ultraviolet photon splits into two red photons, but this does not mean that an ultraviolet photon is composed of two red ones. It only means that the energy of the two red photons equals the energy of the ultraviolet photon. We can say that energy not only changes form but also “dresses” itself in different particles of being. As for the particles themselves, they undergo transformations – their creation and annihilation For example, during beta decay, a free neutron transforms into a proton, an electron, and an antineutrino.
The quantization of the world, originally motivated by the division of matter, adopted the idea of particles as the fundamental building blocks of our universe. Yet quantization is observed for all physical quantities. In practice, this means that for different systems, some quantities can only take certain values. These values are not always integer multiples of some basic quantum of the given quantity. For example, the allowed energy levels of the hydrogen atom are multiples of ( n = 1, 2, 3, …) of the energy –13.6 eV, and the emitted photons have frequencies proportional to the differences between these energies. The quantization of atomic energy, as described by the Schrödinger equation, solved the problem of classical physics equations.
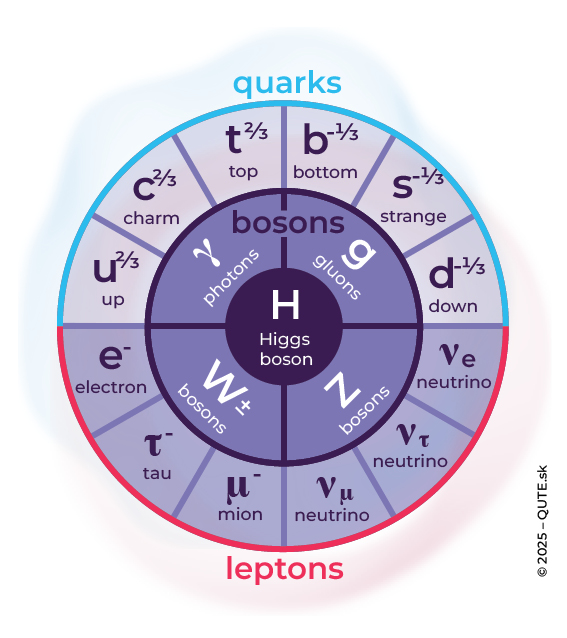
Table of elementary particles according to the so-called Standard Model. The outer circle consists of fermions with spin. The inner circle contains the ‘interaction’ bosons with unit spin, and the Higgs boson at the center has zero spin. The superscript indicates the electric charge.
Measuring Time
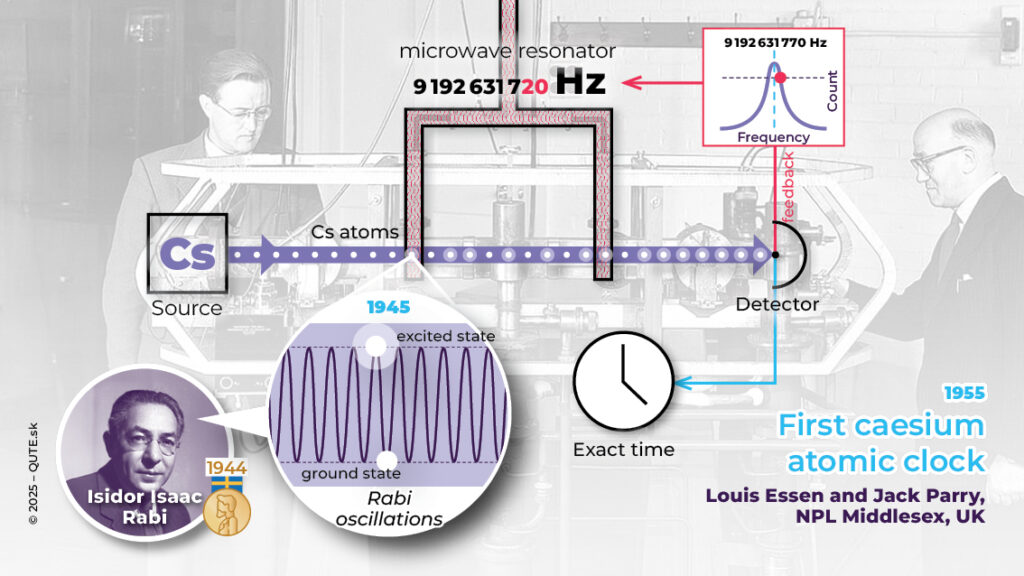
Simplified principle of atomic clocks. In 1945, Isaac Rabi proposed using the oscillation frequencies of atoms to maintain the stability of time measurement. The first caesium atomic clock was built in 1955 by Louis Essen and Jack Parry in England.
Since 1967, the definition of the second has been based on the frequency of photons associated with the transition between two energy levels (the hyperfine structure of the ground state) of the 133Cs atom (in the absence of external fields). A cesium atom in its ground state is excited in a microwave resonator with a frequency of 9192631770 Hz (it absorbs a microwave photon). In atomic clocks, caesium atoms pass through such a resonator, and a detector counts how many of them become excited. If the resonator’s frequency is exact, all atoms are excited. If it is not precise, the number of excited atoms decreases relatively quickly, allowing us to adjust the radiation frequency. This frequency is used to measure time (it is the “tick” of the clock), and caesium atoms act as the tool that keeps this frequency stable.
Caesium atomic clocks have been determining accurate time worldwide for over 60 years. They have an error of only one second in several million years (a few nanoseconds per day). The precise synchronization they provide is crucial for functioning high-speed internet. Without the accuracy of atomic clocks, global navigation systems would have precision in kilometers rather than meters.
The next generation of so-called optical atomic clocks uses energy levels associated with visible-light photons, improving the accuracy of caesium clocks by up to 100 000 times. In addition to refining the definition of the second, these clocks will also enable more precise navigation in space.
Author of the article: Mário Ziman, Institute of Physics, Slovak Academy of Sciences, Bratislava
Illustrations: Diana Cencer Garafová, QUTE.sk – Slovak National Center for Quantum Technologies
Translation: Gabriela Kotúčová
Image Source: wikipedia public domain



